The AI-MultiTrack project, a Eurostars-awarded initiative, is set to revolutionize abdominal injury assessment by integrating automated MRI and CT co-registration directly into BMD Software’s PACScenter. This collaboration aims to tackle the 1.4 million annual deaths caused by abdominal tumors. This partnership between BMD Software, SYCAI, and ULSRA (Unidade Local de Saúde da Região de Aveiro) focus on revolutionizing abdominal diagnostics. The project develops a pioneering medical image co-registration system that streamlines the detection and monitoring of abdominal injuries. Therefore, it will be possible to ensure higher diagnostic accuracy through advanced technological integration. Figure 1 displays PACScenter platform featuring the SYCAI AI-MultiTrack integration.
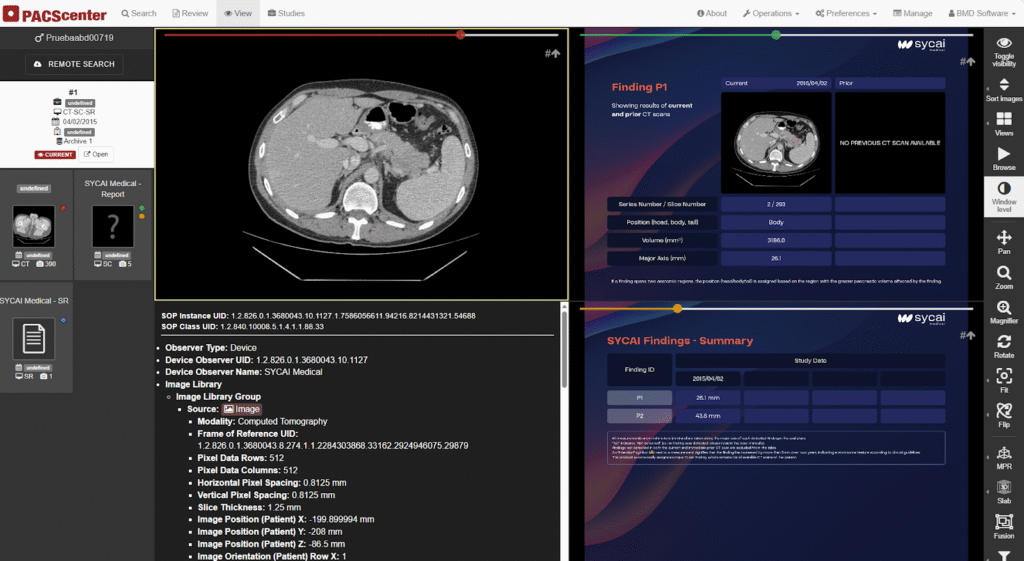
Fig. 1 – PACScenter’s web-native interface streamlines abdominal workflows by integrating SYCAI’s automated lesion detection and historical comparison tools.
The Challenge: Overcoming Abdominal Diagnostic Complexity
Abdominal solid organ tumors affect, for example, the liver, pancreas, and kidneys. They are responsible for more than 1.4 million deaths per year globally. Depending on the types and locations, it is not always easy to achieve an accurate characterization of the lesions. Depending on the types and locations, a timely diagnosis of malignancy remains a significant challenge.
AI-MultiTrack emerges to address this important limitation. It will be using advanced Artificial Intelligence (AI) algorithms to analyze Magnetic Resonance Imaging (MRI) and Computed Tomography (CT) images, allowing for:
- Earlier and more accurate diagnoses.
- Improving the therapeutic approaches.
- Increase patients survival rates.
How PACScenter Integrates AI for Abdominal Diagnostics | Technological Innovation and Clinical Integration
BMD plays a central role in integrating this technology into its PACScenter platform. This evolution will allow the radiologist to identify and monitor lesions over time, automatically comparing the patient’s history on PACScenter.
“This feature not only increases diagnostic accuracy but also makes the system highly attractive to markets with a shortage of specialists, such as Central Asia, the Middle East, Latin America, and Africa”, says the company’s CEO.
ULSRA provides the clinical validation, data ecosystem, and expertise for the project. PACScenter offers easy integration within ULSRA, since it is a 100% web-native platform, the system requires no heavy local installations. By testing the tool’s impact on hospital workflows, the institution ensures the AI serves as a dedicated right-hand for the radiologist.
A New Standard in Oncological Imaging in Portugal
The initial impact of AI-MultiTrack will be felt in the Aveiro region. However, the project’s ambition reaches a national and international scale. AI-MultiTrack transforms the complexity of abdominal diagnosis into a digitally assisted task. Therefore, the project aims to establish a new standard of excellence in oncological imaging.
ULSRA and BMD with this partnership reinforce their position at the forefront of technological innovation. Additionally, it actively contribute to global scientific advancement by sharing results that promise to redefine the future of healthcare by embracing the current trend of so-called precision medicine.

This work was partially supported by Portugal 2030 (P2030) via the Centro Regional Programme (Centro 2030) and the European Regional Development Fund (FEDER) under grant CENTRO-01-0247-FEDER-02590100. The AI-MultiTrack project is also funded by the Eurostars-3 Programme, supported by the Eureka Network and the European Union (Project ID: 7402).
